A handheld device that provides targeted, long-lasting pain relief through Cryoneurolysis
Cryoneurolysis is the freezing of the signal-carrying parts of a nerve
The iovera° system creates a “cold zone” that blocks nerve signals and immediately reduces pain—without the use of drugs*:

Targeted cold is applied directly to a peripheral nerve

The iovera° treatment does
not include the injection of opioids

Pain relief can last up to 90 days per treatment1

iovera° uses nitrous oxide and the body’s own fluid to create a precise cold zone under the skin cold enough to immediately prevent the nerve from sending pain signals without causing damage to the surrounding structures

The effect on the nerve is temporary, providing pain relief until the nerve regenerates and function is restored
Cryoneurolysis with iovera° provides targeted pain relief without permanently damaging nerves
1
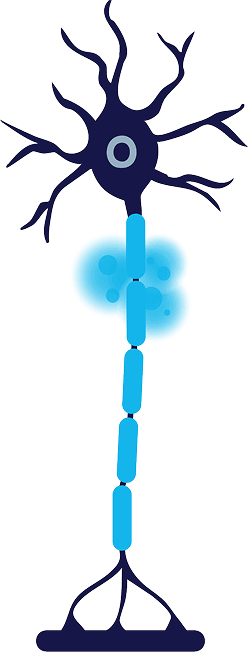
Treatment
Cold zone is created around the targeted nerve, reaching a temperature of -88°C
2
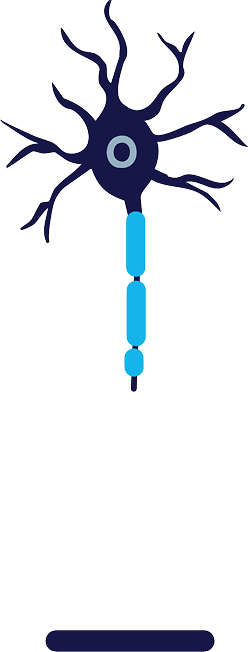
Degeneration
Cold zone causes degeneration of axon and myelin sheath, temporarily blocking nerve signals
3
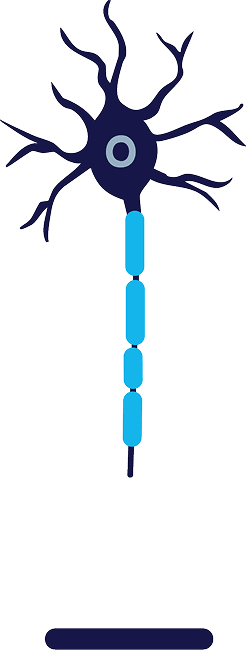
Regeneration
Following treatment,
the axon regenerates
at about 1 to 2 mm/day2
4
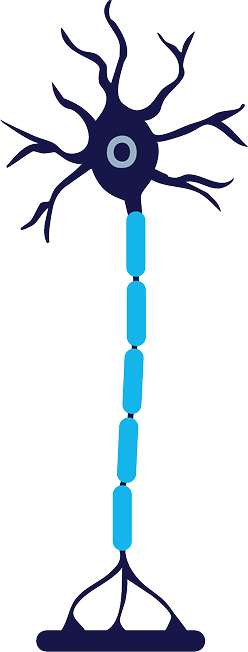
Reinnervation
The axon and myelin sheath are fully regenerated, and nerve signaling is restored
*iovera° therapy does not treat the underlying cause of the pain; timely remediation is necessary to address the root cause.
- References:
- 1. Radnovich R et al. Osteoarthritis Cartilage. 2017;25(8):1247-1256.
- 2. Evans PJ et al. Br J Anaesth. 1981;53(11):1121-1127.